System Requirements
System Requirements Document (SRD) for project-200840c5
1. Introduction
This document outlines the system requirements for project-200840c5, a simple and interactive educational application designed to explain the concept of the galvanic cell (خلية جلفانية). The project is intended as a school project for سيف in Qatar (QA) and focuses on delivering a straightforward, visually engaging, and educational experience.
The application will provide an easy-to-understand explanation of how chemical reactions in a galvanic cell generate electricity, using animations, audio explanations, and a simple interactive test.
2. System Overview
The primary goal of project-200840c5 is to create a basic educational application that explains the galvanic cell in a clear and engaging manner. The application will feature:
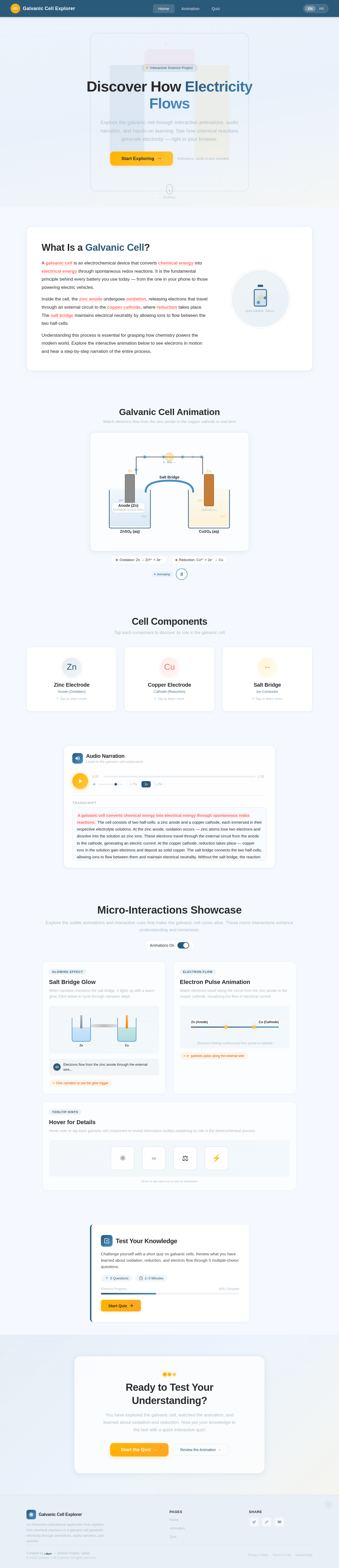
- A visual animation to demonstrate the movement of electrons between zinc (anode) and copper (cathode).
- Audio narration to explain key concepts such as oxidation, reduction, and the role of the salt bridge.
- A simple interactive quiz to reinforce learning.
- A clean and intuitive user interface tailored for students.
The application will be lightweight, accessible on both web and mobile platforms, and optimized for educational use.
3. Functional Requirements
The following are the functional requirements for project-200840c5:
- As a User, I should be able to view an animated explanation of the galvanic cell.
- As a User, I should be able to listen to an audio narration explaining the galvanic cell's components and processes.
- As a User, I should be able to interact with a simple quiz to test my understanding of the galvanic cell.
- As a User, I should be able to navigate between sections easily.
4. User Personas
4.1 Student
- Description: The primary user of the application. A student who wants to learn about the galvanic cell for a school project.
- Goals: Understand the concept of the galvanic cell through visuals and audio, and test their knowledge with a quiz.
- Technical Proficiency: Basic familiarity with mobile or web applications.
4.2 Teacher
- Description: A secondary user who may use the application to demonstrate the concept in a classroom setting.
- Goals: Use the application as a teaching aid to explain the galvanic cell.
- Technical Proficiency: Moderate familiarity with mobile or web applications.
5. Visuals Colors and Theme
The visual theme for project-200840c5 will reflect a scientific and educational aesthetic. The color palette is designed to be engaging yet professional, with a focus on clarity and readability.
Color Palette:
- Background: #F5F9FF (Soft Sky Blue)
- Surface: #FFFFFF (Pure White)
- Text: #2A2A2A (Dark Charcoal)
- Accent: #FF6F61 (Bright Coral Red)
- Muted Tones: #B0BEC5 (Soft Gray-Blue)
This palette ensures a clean and modern look, suitable for an educational application.
6. Signature Design Concept
The signature design concept for project-200840c5 is an interactive animated diagram of the galvanic cell on the homepage.
Key Features:
- Dynamic Animation: The homepage will feature a visually engaging animation of the galvanic cell. Users will see electrons moving from the zinc anode to the copper cathode, with labels appearing dynamically to explain each component (e.g., "Anode," "Cathode," "Salt Bridge").
- Interactive Elements: Users can tap or click on components (e.g., zinc, copper, salt bridge) to hear a brief audio explanation of their role in the galvanic cell.
- Audio Narration: A voiceover will play automatically, synchronized with the animation, explaining the oxidation and reduction processes in simple terms.
- Micro-Interactions: Subtle animations (e.g., glowing effect on the salt bridge when it is mentioned) will make the experience more immersive.
- Quiz Transition: At the end of the animation, a button will appear inviting users to take a short quiz to test their understanding.
This design ensures that the application is not only educational but also memorable and engaging.
7. Non-Functional Requirements
- The application must load within 3 seconds on standard mobile and desktop devices.
- The audio narration must be clear and synchronized with the animation.
- The application must be responsive and work seamlessly on both web and mobile platforms.
- The quiz functionality must store user answers temporarily for the session but not require persistent storage.
8. Tech Stack
The following technologies will be used to develop project-200840c5:
Frontend:
- React for the web interface.
- React Native for the mobile application.
Backend:
- Python with FastAPI for serving content and managing quiz logic.
Database:
- MySQL for storing quiz questions and answers (if needed).
AI Tools:
- Google Nano Banana for generating any required educational images.
Local Orchestration:
- Docker for containerization.
- docker-compose for managing local development environments.
9. Assumptions and Constraints
Assumptions:
- The target audience has access to either a smartphone or a computer.
- The application will be used primarily in an educational setting.
- Audio narration will be recorded in Arabic to cater to the local audience in Qatar.
Constraints:
- The application must be lightweight and not exceed 50 MB in size.
- Development must be completed within the timeframe of the school project.
10. Glossary
- Galvanic Cell (خلية جلفانية): A device that converts chemical energy into electrical energy through redox reactions.
- Anode: The electrode where oxidation occurs (zinc in this case).
- Cathode: The electrode where reduction occurs (copper in this case).
- Salt Bridge: A device that maintains charge balance by allowing the flow of ions.
- Oxidation: The loss of electrons.
- Reduction: The gain of electrons.

No comments yet. Be the first!